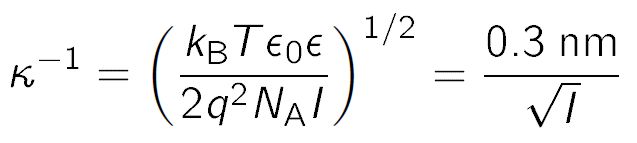![SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4] SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]](https://cdn.numerade.com/ask_images/6fe865672fb949d8969b3329b257473c.jpg)
SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]

Solved: Chapter 12 Problem 11P Solution | Fluid Mechanics For Chemical Engineers With Microfluidics And Cfd 2nd Edition | Chegg.com

The-dependence of the Debye length D in a plasma of temperature k B... | Download Scientific Diagram

SOLVED: a) Show that the Debye length can be approximated by (kT / (4Ï€e^2n0))^1/2, where T is the temperature in Kelvin and n0 is the number density in m^-3. b) Show that

Relation between the Debye length, ECV technique coordinate resolution,... | Download Scientific Diagram
Nanofluidic osmotic power generators – advanced nanoporous membranes and nanochannels for blue energy harvesting - Chemical Science (RSC Publishing) DOI:10.1039/D1SC03581A

SOLVED: HCl is a covalent molecule. The covalent bond in HCl has 24 % ionic character. The experimental dipole moment of this molecule is found to be 1.50 Debye. Calculate the bond

SOLVED: BACK-OF-THE-ENVELOPE 9.4 What is the Debye length of a microfluidic system with 10mM NaCl salt solution dissolved in water? What about for 10uM? Since NaCl is a symmetric electrolyte, we can








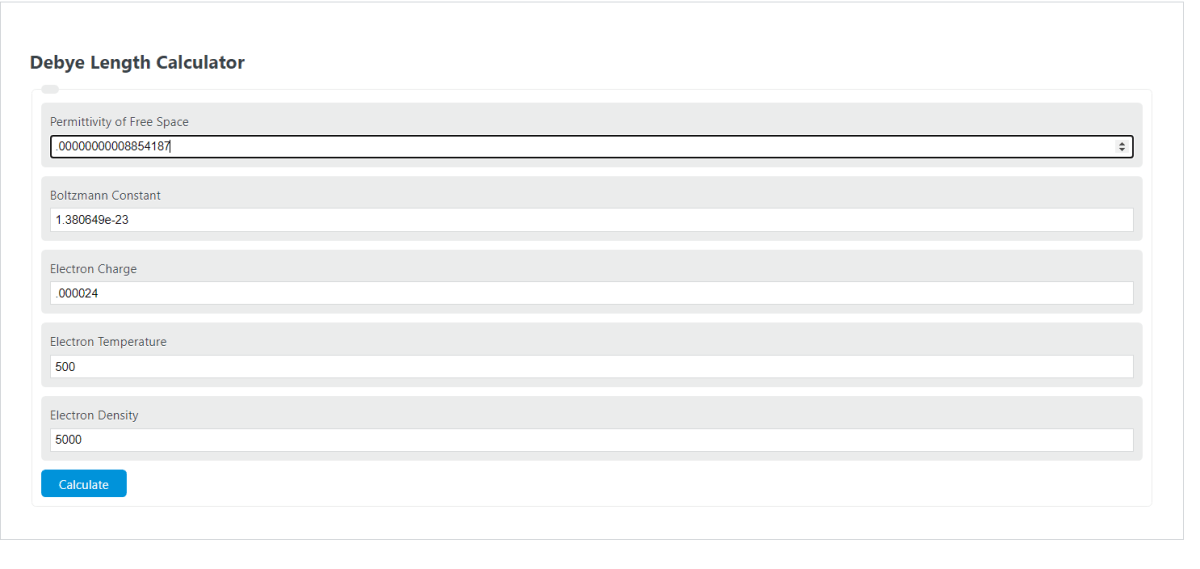
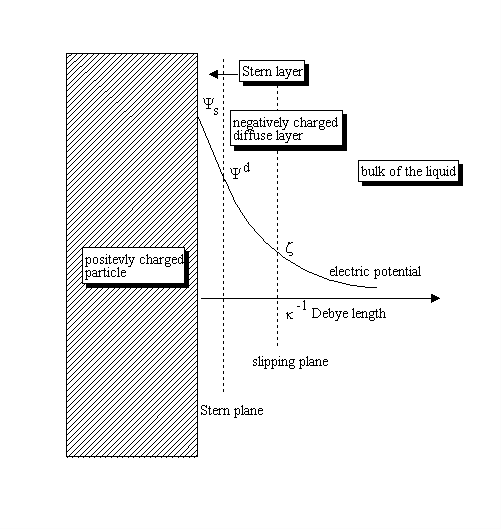


![Solved where -1 ɛɛk]T к = Ze² Po Debye length if written as | Chegg.com Solved where -1 ɛɛk]T к = Ze² Po Debye length if written as | Chegg.com](https://media.cheggcdn.com/media/bbd/bbd52633-2636-4663-aa0f-a4b35e6852ec/php413gmk.png)