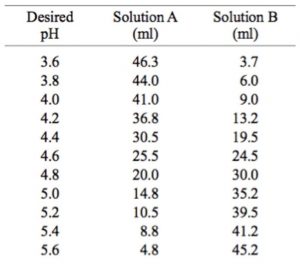
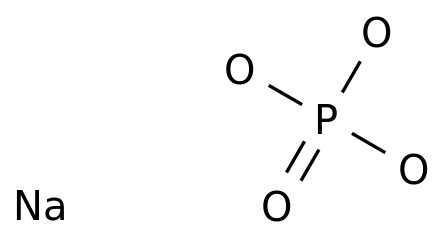
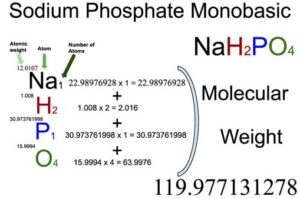
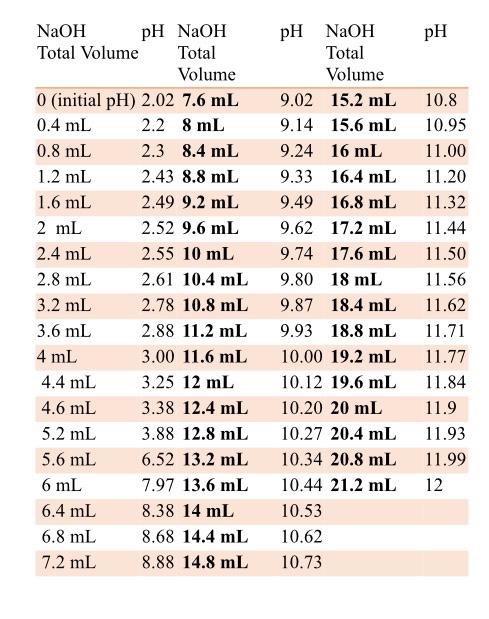
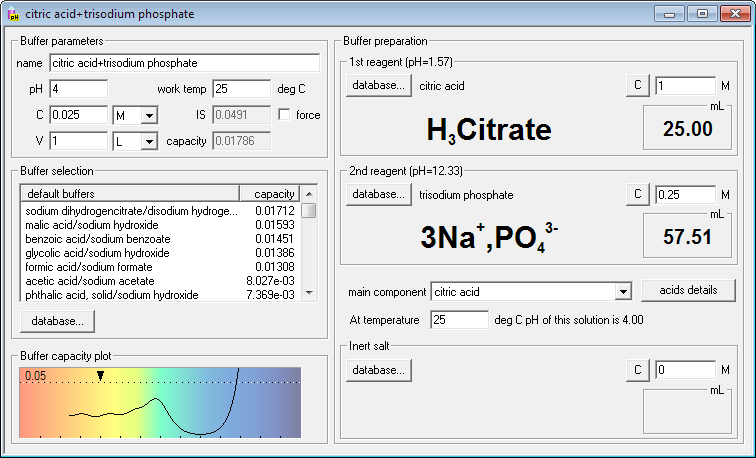
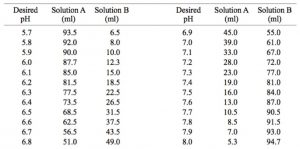
SOLVED: And, if necessary, adjust the pH to 7.4 (using either the 0.1 M HCl or 1.0 M NaOH solutions). Adjust the final volume to 100 mL with DDW. Save this solution

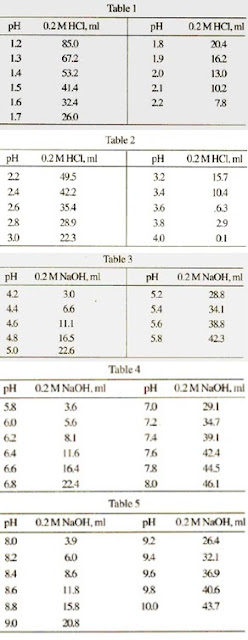
SOLVED: Calculate the Molar Extinction Coefficient (€) at 351 nm for aquocobalamin in 0.1 M phosphate buffer, pH = 7.0, from the following data which were obtained in a 1 cm cell: Solution Cx10^-8 A 2.23 100 27 B 1.90 100 32.

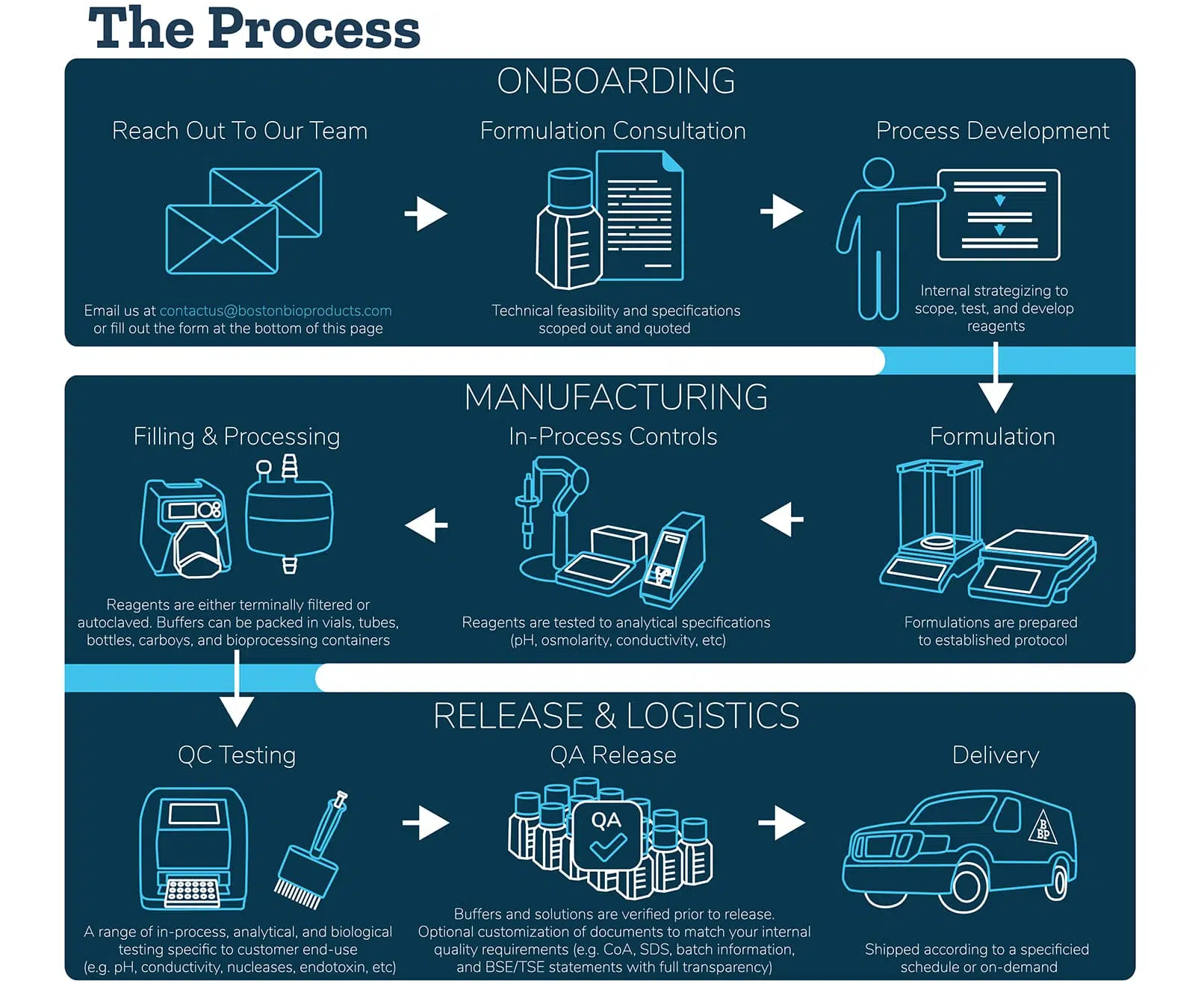
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability



![Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/01/sodium-phosphate-dibasic-dihydrate-molecular-weight-calculation-300x186.jpg)